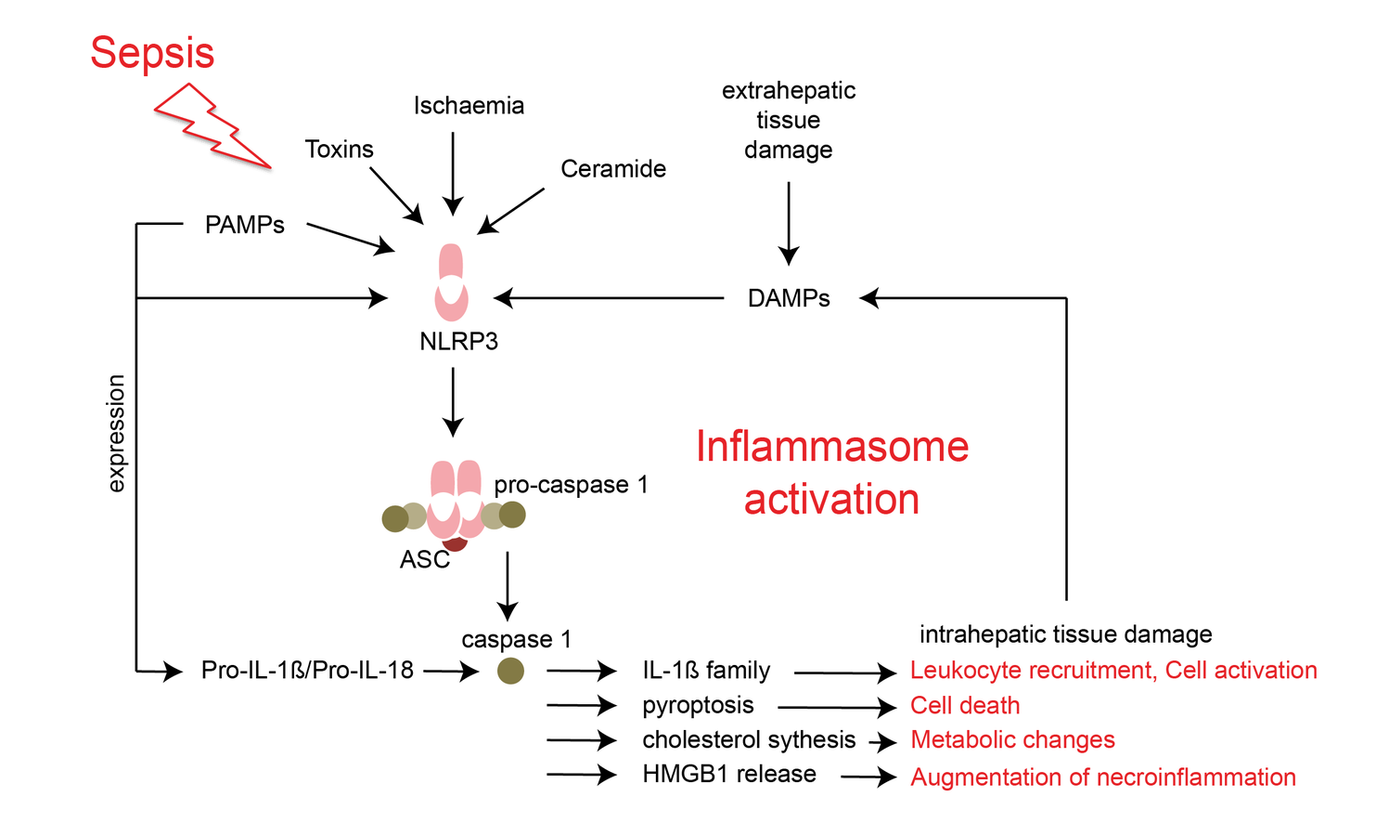
The inflammatory immune response during sepsis is a double-edged sword, which supports pathogen elimination on one hand but may induce and aggravate organ failure on the other hand. The activation of inflammasome complexes is crucial for responding to invading pathogens during sepsis but mounting evidence suggests that unrestricted inflammasome activation plays a central role in perpetuating organ damage in a variety of diseases. Hepatic activation of NLRP3 induces inflammation, fibrosis, and cell, but its regulation in septic liver injury is poorly understood.
In this project we will investigate the regulation of the NOD-like receptor NLRP3 during septic liver injury and its consequences on hepatic inflammation, metabolism, and cell death. We will study inflammasome activation in different cell types using the model of polymicrobial abdominal sepsis. The use of molecular multimodal imaging will allow to investigate the consequences of organ injury in a fast and label-free fashion over large areas of the liver in real-time. In addition, we will investigate, whether - and what kind of - soluble factors mediate inflammasome activation in patients with septic liver injury.
The overall aim of the project is to understand time- and cell-specific regulation of inflammasome activation during the course of sepsis in order to provide the opportunity of delivering targeted anti-inflammatory strategies to improve the outcome of patients with septic liver injury.